Learn about the cause and effects of cystic fibrosis
Learn MoreFind out how cystic fibrosis is treated at home and in the hospital
Learn MoreDiscover the latest research about cystic fibrosis and therapies under development
Learn More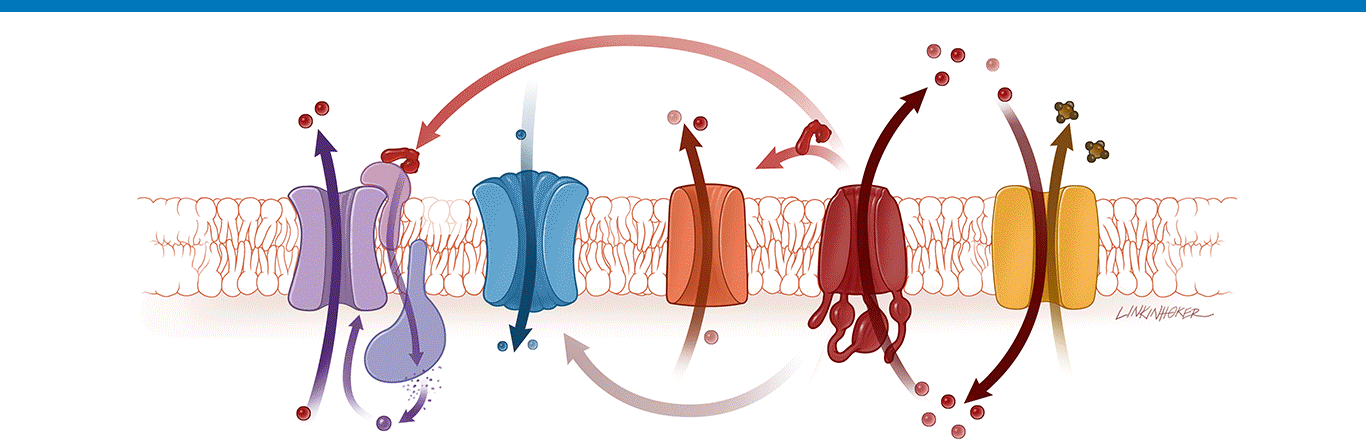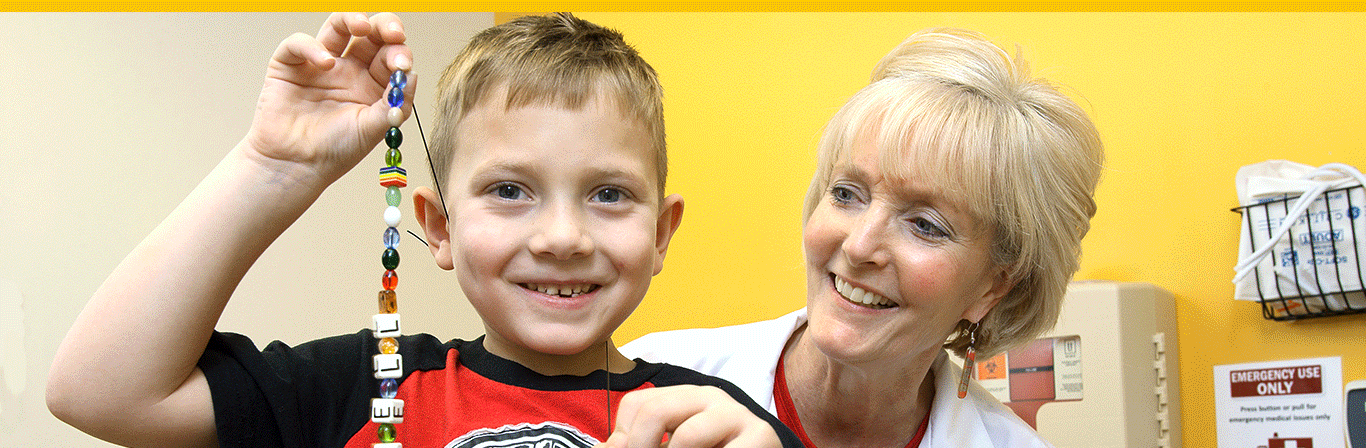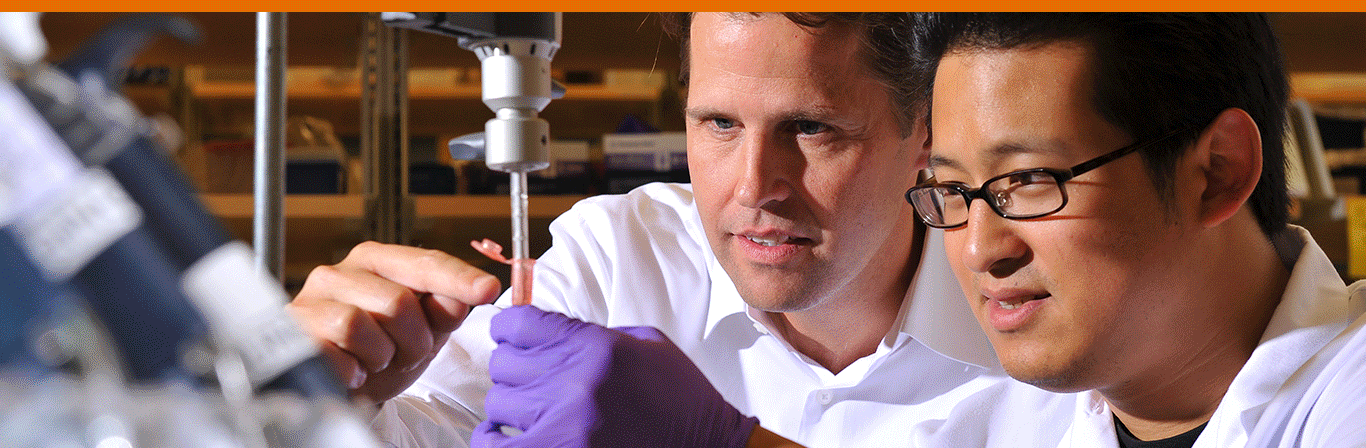
Learn about the cause and effects of cystic fibrosis
Learn MoreFind out how cystic fibrosis is treated at home and in the hospital
Learn MoreDiscover the latest research about cystic fibrosis and therapies under development
Learn More